by Julia Seibert
Draconian: an adequate way of describing the headaches involved with sending something into space. Escaping the iron clutches of Earth’s gravity requires powerful rockets, meticulous planning, and a good bit of luck. It also requires rocket propellant. Lots of it. For the payload to reach orbit, up to 96% of a rocket might need to consist of propellant, which the comparatively feather-light vehicle burns through in a matter of minutes.
Despite these extreme ratios, rocket fuel cost makes up only a tiny portion of a launch cost, which can run anywhere from tens of millions to even billions. The bulk of the price comes from developing, building, and operating the rocket itself, which is often simply thrown away after its launch. A recent rise in reusable rockets has been slashing these costs, but will propellant prices remain negligible?
Henceforth, the term “rocket propellant” will be used instead of “rocket fuel” to ensure precise terminology. Rocket propellant is composed of two primary components: fuel and oxidizer. The fuel component provides the energy source, while the oxidizer supplies the necessary oxygen for combustion.
Table of Contents
ToggleWhat Makes Rocket Propellant So Expensive?
Contrary to what one might think, rocket propellant cost is not that high. Sure, a $200,000 tanking cost for just one launch might seem jaw-dropping for the average spectator, but it pales in comparison to the millions the rest of the undertaking costs. According to Henry Spencer, a founding member of the Canadian Space Society and Fellow of the British Interplanetary Society, ‘fuel is cheap – it’s the hardware and people that cost you… current launch costs are dominated by salaries, not fuel prices. Simply put, a few hundred thousand dollars for propellant are peanuts for the space industry.
According to moving average US defense prices, LH2 costs around $6.1 a kilogram (though standard prices by the Defense Working Capital Fund indicate a much higher price of $30.5/kg). RP-1 presents a cheaper option at $2.3/kg. The document lists CH4 at $8.8/kg, while LOX comes in at $0.27/kg. Meanwhile, solids cost around $5/kg as of 2008, while hybrid propellants are significantly more expensive. HTPB (which is not listed in the document but discussed in this 2008 study by Purdue School of Aeronautics and Astronautics) comes in at $8/kg and hydrogen peroxide at $10.36/kg. Hydrazine tops the chart at up to around $75.8/kg.
Examples of Rocket Propellant Cost
SpaceX Falcon 9
Both stages of SpaceX’s workhorse Falcon 9 rocket guzzle about 312,200kg of LOX and 186,006kg of RP-1 throughout a flight. That results in a propellant cost of about $512,108, an overshot of Musk’s 2011 ballpark of $200,000 a launch (still, exact costs are rarely made public by companies; these estimations are based on the defense costs listed above and may be different in reality). This number also does not include helium, which is used to backfill the tanks and keep them pressurized, but exactly how much of that is used is unclear.
You may also like: Top 6 SpaceX’ Goals

NASA Space Shuttle
The Shuttle used around 729,007kg of various liquid propellants, which set NASA back $1,380,000 in 2001. They included 103,328kg of LH2, 612,350kg of LOX, 492kg of hydrazine, 4,899kg of monomethylhydrazine (MMH), and 7,938kg of nitrogen tetroxide. In addition to the liquids, the Shuttle had two solid rocket boosters strapped to its sides to provide additional thrust during launch. Each of these contained over 450,000kg of propellant, costing roughly $4.5 million in 2008 dollars. In total, propellant would have cost roughly $4,658,043 in 2008 for one launch. Again, this is practically nothing compared to the $450 million average launch cost, which rises to $1.5 billion per launch when considering the entire budget.
European Ariane 5
Europe’s one and only large-scale rocket consists of two liquid-fueled stages, plus two solid rocket boosters. The first stage runs on 132,270kg of LOX and 25,840kg of LH2, and its second stage devours approximately 12,220kg of LOX and 2,380kg of LH2. Together, that would set Arianespace back about $211,154, according to the 2023 US defense prices. Then there are the two solid boosters, each consisting of 475,000kg of propellant, costing around $4.75 million together. The entire propellant cost therefore lies at almost $5 million. Ariane 5 costs $165-200 million to launch, so propellant prices are once again negligible.
You may also like:
- How Much Does It Cost to Launch a Rocket?
- Top 10 Rocket Launch Companies To Look For in 2023
- The 20 Best Movies About Space to Watch In 2023
Factors Affecting Rocket Propellant Cost
Some of the above market prices might seem like a steal, but they are not final. Several factors further influence propellant costs and launch prices, including the handling of the chemicals. For example, hydrazine is expensive as is, but its toxicity and volatility mean that managing it further drives prices up. Similarly, dealing with the annoyances of LH2, including the installation and upkeep of complex seals and insulation, are likely to increase the overall costs of using this fuel. By contrast, CH4 is more expensive, but can be stored at a similar temperature to LOX and is less prone to escaping through gaps, which could make it cheaper compared to LH2. This becomes important for rapid reusability, for which the more efficient yet fickle LH2 is not as well suited to as other fuels. On the other hand, while the partially reusable Falcon 9 uses RP-1 as fuel, such carbon-based substances coke up the engines and shorten their lives. Frequently reusing rockets (and engines) decreases launch costs, so a more expensive fuel like CH4 could pay off in the long run.
In addition, if space colonization plans ever materialize, producing rocket propellant in-situ (away from Earth using locally available materials) will be vital. More complex chemicals such as those used by hybrid engines may not be the best option here, whereas hydrogen and oxygen could be extracted from water, and methane synthesized from carbon dioxide on Mars. If such processes become a reality, they might be cheaper than hauling extra propellant around the solar system.
Types of Rocket Propellants
Some basics before getting into the nitty gritty of different rocket propellants: as explained in detail by Everyday Astronaut, the basic function of a rocket is to expel as much mass as possible out of one end to get it moving in the opposite direction. A great way to do this is to create as much pressure and heat as possible in the combustion chamber of the engine, which then converts it to kinetic energy and propels the rocket upwards. Generating that energy is the primary job of rocket propellants, though these can take different forms depending on the mission.
However, physics throws a spanner in the works. As laid out in Russian scientist Konstantin Tsiolkovsky’s landmark rocket equation, more payload requires more energy, which means more propellant, which in turn increases the mass, meaning yet more propellant. This vicious cycle illustrates the reason why so much propellant is needed to launch something into orbit, and engineers work tirelessly to squeeze these ratios to the limit to allow for maximum payloads. Therefore, only the most efficient propellants make the cut for rocket science.
Common Combustible Liquid Propellants: Cryogens, Petroleum, and Hypergolics
Liquid propellants are among the most frequently used in the industry. They consist of two parts: a fuel and an oxidizer, the latter of which is used to help the fuel burn in space, where no oxygen is present. Before launch, they are stored in separate tanks within the rocket. At go time, they then meet in the combustion chamber (or in the plumbing leading up to it, depending on the engine cycle), where they ignite and create the energy needed to propel the vehicle forwards. Depending on the mission, their flow can be throttled or turned on and off, making them suitable for complex missions or reusable rockets.
Cryogenic propellants are found in some of the world’s highest-mass launch vehicles, including NASA’s Space Shuttle, Space Launch System, and the upper stages of the Saturn V moon rocket, as well as Arianespace’s Ariane 5 and SpaceX’s still-prototypical Starship. They consist of gases – most commonly liquid oxygen (LOX) as oxidizer and liquid hydrogen (LH2) as fuel – chilled to frigid temperatures to keep them in their liquid state. The reason these are often used to launch heavy payloads is their efficiency, which rocket scientists refer to as specific impulse. This basically translates to how quickly the mass can be tossed out of the engine, for which a low molecular weight and high exhaust temperatures are key, and cryogenic propellants happen to have exceptional values for both.
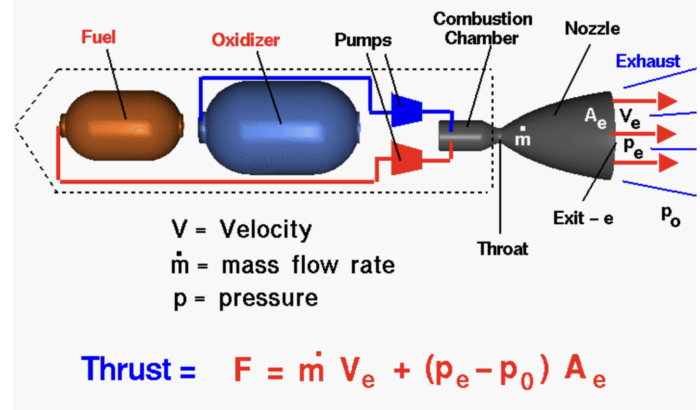
The LOX/LH2 combo, of which NASA is a big fan, has the highest specific impulse of them all, meaning less of it needs to be packed. Still, LH2 is somewhat of a diva, requiring temperatures of -253 degrees Celsius to stay liquid, as opposed to the -183 degrees for LOX. This, combined with its low density, requires enormous insulating tanks that add weight to the rocket. In addition, LH2 likes to leak out of even the tiniest of cracks, which can lead to explosions: a generally unfavorable event when it comes to rockets. Therefore, SpaceX’s Starship, which aims to launch in quick succession, uses liquid methane (CH4), which is slightly less efficient than LH2, but is denser and can be stored at a similar temperature to LOX, requiring less insulation.
Petroleum is another viable liquid propellant option and has been used on the first stages of Saturn V, Atlas, and Delta II, as well as SpaceX’s Falcon 9. The preferred petroleum for rockets is RP-1, a highly refined jet fuel. Though it can be stored at room temperature, it is usually used in combination with LOX, so some insulation is still required. Plus, SpaceX slightly chills their RP-1 to increase density.
Still, while less efficient than cryogens, RP-1 is cheaper and easier to handle than LH2 or even CH4. Its biggest issue is that being a fossil fuel, RP-1 is quite messy. It leads to coking, or soot, on the engines, shortening their lifespan. Rockets guzzling RP-1 also emit particles of black carbon during flight, which contributes to warming up the atmosphere. While all rockets come with environmental concerns, black carbon is an extra kick in the shins for the climate.
Yet another vein of liquid propellants frequently used in the industry are hypergolics, which ignite on contact with each other. They often consist of some form of hydrazine (fuel) and nitrogen tetroxide or nitric acid (oxidizer). These can be kept at room temperature, meaning they do not require the extensive insulation antics needed for their cryogenic cousins, and can therefore be stored for much longer. Though less efficient than cryogens or petroleum, their no-fuss combustion mechanism, storage time, and quick deployment makes them perfect for maneuvering in space, and are often used by satellites, deep space probes, and reaction control systems of human-carrying vehicles such as the Apollo Command and Service module, the Space Shuttles, and the SpaceX Dragon spacecraft.
However, hypergolics are dubbed the Devil’s Venom for a reason. In addition to exploding when improperly stored, they are toxic and carcinogenic to humans coming into contact with them, and produce a poisonous gas when combusted. Their use has resulted in numerous accidents, including the Nedelin catastrophe, the deadliest ever rocketry accident. So, while hypergolics can and have been employed as propellant for launching from Earth, their use is mostly confined to space.
Solid Propellants
Solid propellants are exactly what they sound like. They look like cement-colored cake, and consist of pre-mixed chemicals that need only be ignited at takeoff to produce staggering amounts of thrust. These mixtures might be homogenous, consisting of only one or two chemicals, which are less efficient but harder to trace, making them ideal for missiles. Space-bound rockets use composite or heterogenous solids consisting of a fuel and oxidizer, often aluminum and ammonium perchlorate, and some additional chemicals for binding or higher burn rates. Either option is easy to store and deploy, with just one small problem: after ignition, there is no way to turn them off. The flame, which starts in a hollow center channel of the cake, burns from the inside out and eats its way through the propellant until there is none left. The result can best be described as an enormous firecracker, but a very effective one.
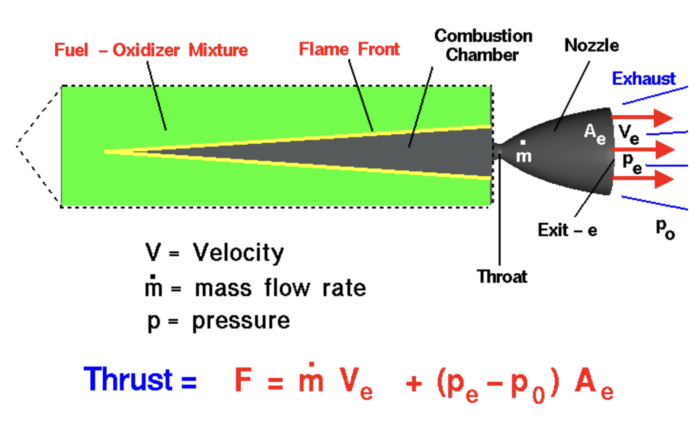
While liquid fuels are generally more efficient, strapping solid boosters onto a liquid-fueled rocket can increase payload capacity without adding too much dead weight, and can be seen on the Space Shuttle, Space Launch System, and the Ariane 5, to name a few. Alone, solids have been used to launch lighter payloads to orbit on rockets such as NASA’s Scout and Northrup Grumman’s Pegasus. Sounding rockets (small vehicles used to send payloads to suborbital altitudes) frequently use solids, too.
Hybrid Propellants
As their name suggests, hybrids are part liquid/gas, part solid propellant systems. Usually, it is the fuel that is solid, with the liquid or gaseous oxidizer kept in a pressure vessel next door. At go time, the fuel is ignited, and oxidizer pumped into the so-called fuel grain, which acts as a combustion chamber. The system’s its efficiency lies somewhere between liquid and solid propellants, though it is used far less than either. Still, hybrids present some significant advantages. They are less finicky than the complex systems used for liquid propellant, while remaining more controllable than solids as they can be stopped and restarted.
Though chemical compositions vary, a synthetic rubber known as hydroxyl-terminated polybutadiene (HTPB) is often used as it is safe to handle, with LOX, nitrous oxide, or hydrogen peroxide as oxidizer. Scaled Composite’s SpaceShipOne, a spaceplane that won the $10 million Ansari X-Prize in 2004, used a hybrid motor with HTPB and nitrous oxide, though the latter caused a deadly accident in 2007 during the development of a successor spacecraft. Today, Virgin Galactic’s SpaceShipTwo uses an upgraded version of the hybrid motor.
Monopropellant
Monopropellants – you guessed it – involve just one propellant in liquid form to power the vehicle. What is not so obvious is that this chemical is run over a catalyst of some sort, after which it decomposes and produces a scorching gas, resulting in thrust. The process does not produce as much energy as the methods listed above, and monoprop engines do not involve any turbopumps or means to accelerate the churning of matter out of the end of the vehicle, so this system is not ideal for escaping Earth’s intense gravity. Still, it is used frequently in satellites for attitude control in space; the German V2 missile even used a monoprop engine to power the turbopump for its main propulsion method. Once again, the fuel is often hydrazine, which is then run over an iridium catalyst. Hydrogen peroxide can also be used as fuel, with platinum or manganese dioxide as possible catalysts.
Looking Ahead: The Future of Rocket Propellant Expense
Right now, the cost of a rocket propellant seems to be the least of any rocket scientist’s problems. The billions invested into getting such a thing to fly in the first place far overshadow a tanking session or two. But will it always be like this? Launch prices have been dropping like crazy in the recent years. Though wildly optimistic predictions of prices as low as $10 million (or even $1 million) for a Starship launch are not yet a reality, the costs of filling a monster like that to the brim with chemicals might someday become significant. The specific cost depends largely on the chemical in question, as energy prices can be volatile and hard to predict. But there is one way for companies to somewhat circumvent this problem: cut out the middleman and produce propellant themselves.
SpaceX, for one, had envisioned this, but have since cancelled plans to build an LNG (liquid natural gas) plant at one of its Texas sites. Still, its rockets might one day run on methane distilled from atmospheric CO2. Similarly, Skyrora has developed a way to turn nonrecyclable trash into Ecosene, a fuel comparable to RP-1. Whether or not launch prices will sink to the extent that such moves will have a significant effect on the cost of spaceflight remains to be seen. But they are certainly worth a shot. In the end, every effort is needed to fight the ruthless costs of rocket science.
If you found this article to be informative, you can explore more current space news, exclusives, interviews and podcasts here.
Featured image: The powerful aluminum-ammonium perchlorate reaction fuels the twin SLS solid rocket boosters. Credit: NASA
Share this article:








